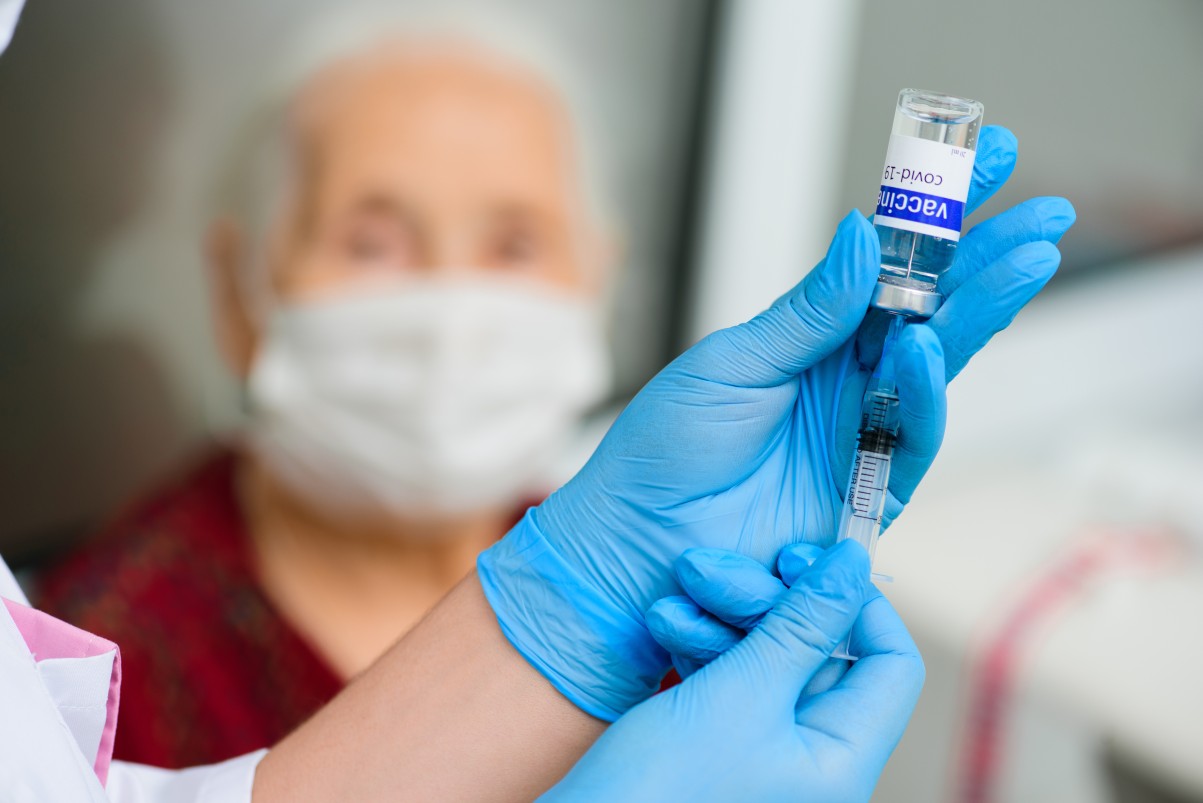
The big news in the fight against COVID-19 this past week was the decision by the US Food and Drug Administration (FDA) and the US Centers for Disease Control and Prevention (CDC) to recommend the vaccine booster shot for some Americans. Are you eligible for this booster and what do you need to know about getting that next shot? Read here for the latest about the COVID-19 vaccination effort.
Before you rush out to get your booster, you need to understand the recommendations surrounding this approval. While there is no central clearinghouse to approve boosters, the CDC and FDA have worked together to set some widely recognized eligibility criteria. Everyone 65 years of age and older is eligible to receive the booster. Adults up to the age of 64 are eligible if they are at a heightened risk of severe COVID-19 because of a pre-existing medical condition. Examples include diabetes and kidney disease.
In addition, women who are pregnant or nursing are also being advised to get their booster shots. Lastly, adults working in healthcare facilities or living in shelters, prisons, or other congregate settings should also get the booster. Other population groups who should strongly consider a booster because of the increased risk include teachers, grocery store workers, and public transportation employees.
What about Johnson & Johnson?
The above recommendations apply to those Americans who received the two-dose Pfizer and Moderna vaccines. Adults who received the first and only dose of the Johnson & Johnson shot two months ago or longer are now also eligible to receive a booster shot. Research shows that the Johnson & Johnson vaccine delivered the least amount of protection of the three shots approved in the US. However, receiving a second dose of the Johnson & Johnson vaccine brings this protection level up to over 90%.
Mixing and Matching Vaccines
The appropriate health oversight organizations have also approved the practice of mixing and matching different vaccines from separate vendors. Known as heterologous boosters, these shots are approved for any of the three currently authorized vaccines in the US. After a thorough review of the evidence, the FDA determined that mixing and matching vaccines provides a greater set of benefits than potential risks.
For example, an individual who originally received the Johnson & Johnson vaccine may choose to receive a Pfizer or Moderna booster. Conversely, the Pfizer and Moderna recipients may also choose to go with the non-mRNA Johnson & Johnson shot.
It should be noted that it is also acceptable to stick with your original vaccine manufacturer when seeking a booster. However, if you experienced a severe reaction with your first vaccine, it is a good idea to check with your healthcare provider to get their recommendations on how to proceed.
Miscellaneous Things to Know
While the Pfizer and Johnson & Johnson boosters are the same dosages as the first shot, the Moderna booster is administered as a half dose.
According to White House Coronavirus Response Coordinator Jeff Zients, all booster shots are free. Although some providers may ask for insurance cards, the vaccine is provided at no cost to Americans regardless of immigration status. These boosters are available for use at more than 80,000 locations in the US. This includes over 40,000 pharmacies and various vaccination sites and medical clinics. According to Zients, about 90% of all Americans live within five miles of a provider offering the vaccine.
The COVID-19 booster can also be administered at the same time as other vaccines. This makes it safe to receive the booster along with your seasonal flu shot, should you choose to do so.
The National Institutes of Health (NIH) has been studying all three of the authorized vaccines to determine when immunity begins to wane. The research demonstrates that all three vaccines deliver a significant immunity boost at about 15 days after the booster shot.
It is also important to remember to bring your original COVID-19 vaccination card with you to your booster shot appointment. Contact your state health department or the place that you received your first vaccination if you have misplaced your card.
What About Kids?
While many older Americans are anxious to get boosted, parents are also anxiously waiting for the approval of the Pfizer vaccine for kids ages 5 through 11. The FDA vaccine advisory committee is meeting on Tuesday to discuss recommending the shot for this age group. Should the FDA give the vaccine the green light, it will go to the CDC for final approval next week.
Most health experts are in agreement that this would make the vaccine likely to be available for widespread distribution by the end of the first week of November. Like the adult version of the Pfizer vaccine, the protocol for children will be a two-shot dosage. The second shot is to be administered three weeks after the first shot.